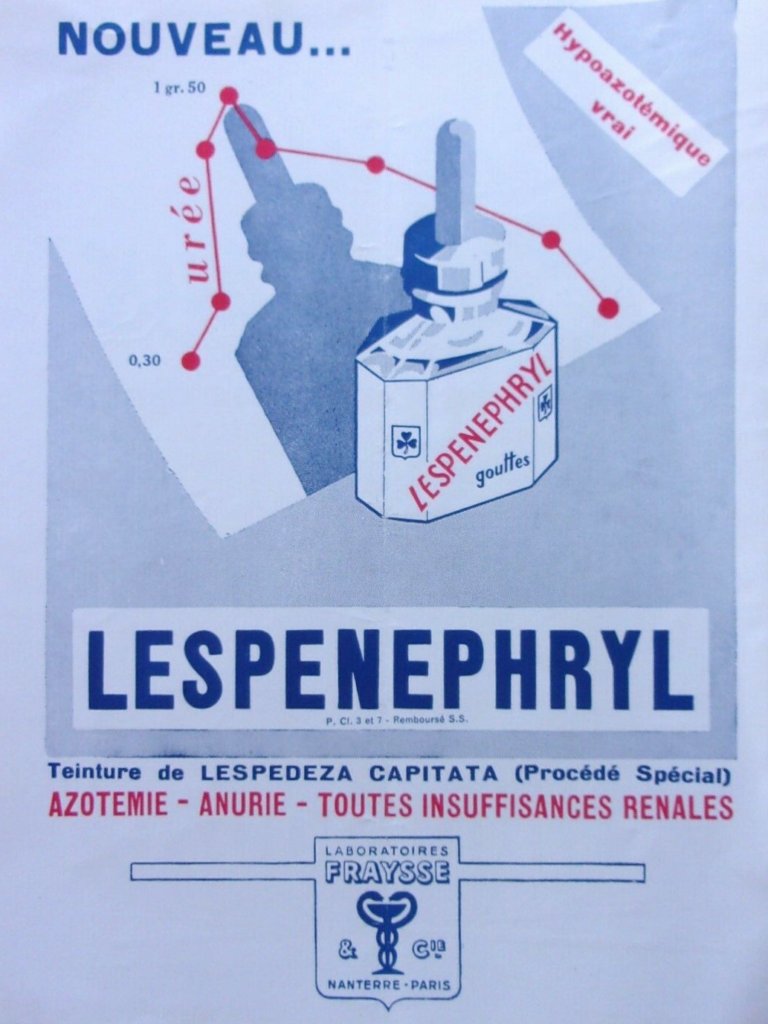
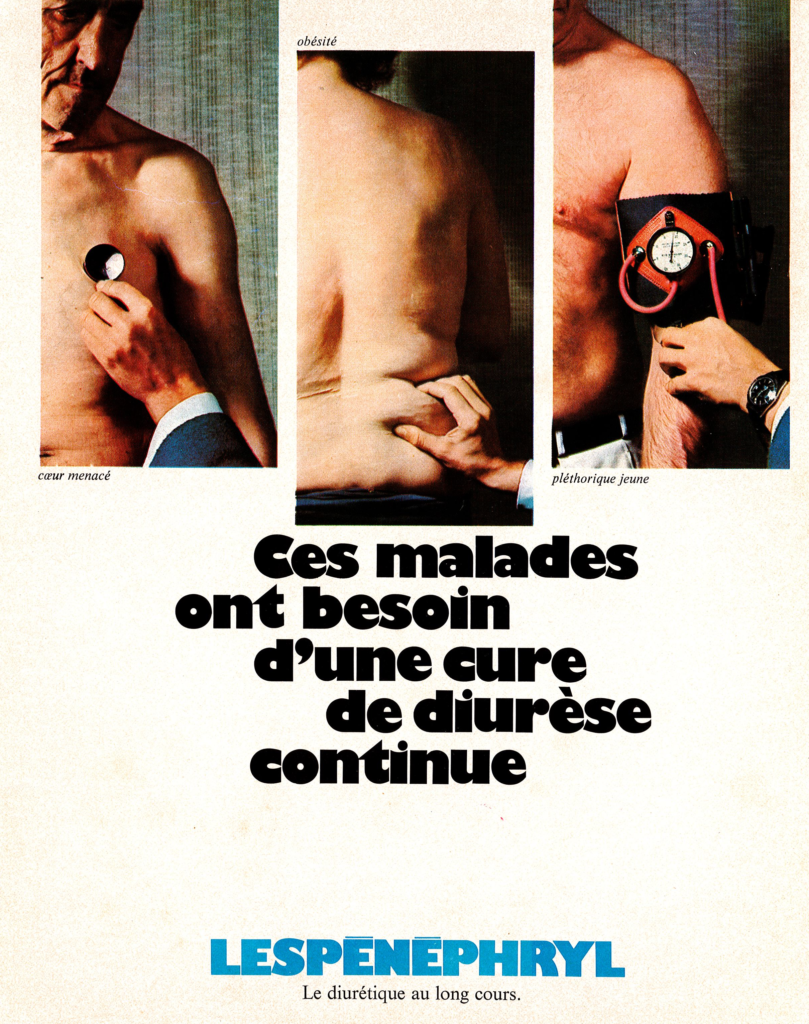
The drug was prescribed in cases of renal insufficiency, notably in patients suffering from heart diseases and/or obesity, for its normolipidemic, hypoazotemic and diuretic actions. Two formulations were proposed originally, an oral liquid form and an injectable solution. Both formulations were derived from extracts of the plant Lespedeza capitata, a flowering plant of the Fabaceae (Leguminosae) family commonly known as round-head bush clover or round-head lespedeza.
The plant, native to eastern North America, is a perennial herb with several medicinal uses. The roots were notably employed to make an antidote for poison or to treat rheumatism. The tincture of L. capitata was also largely used in veterinary medicine to treat cattle, sheep, goats and pigs.
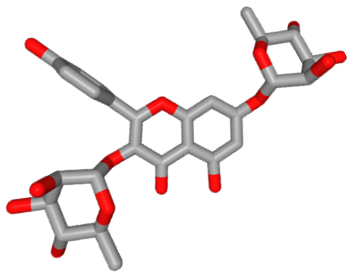
Lespenephryl was removed from the market in 1996, but phyto-therapeutic preparations containing Lespedeza capitata extracts can still be found. Aqueous extracts of L. capitata contain a variety of proanthocyanidins and flavonoids, in particular lespedesin, also called kaempferitrin, or kaempferol-3,7-dirhamnoside, a well-known natural product with diuretic and renal protective properties. Kaempferitrin can be isolated from many plants and is known to ameliorate inflammation in human rheumatoid arthritis but also to inhibit the proliferation of cancer cells, at least in vitro. Kaempferitrin induces useful immunostimulatory effects. After ingestion, this diglycosylflavone derivative is converted into monoglycosyl derivatives and kaempeferol by the human intestinal flora.